Professional Interests – The proper regulation of RNA Polymerase II (Pol II)-dependent transcription—that normally maintains appropriate expression levels of protein-coding genes and non-coding RNAs—is crucial to keep cells healthy and prevent diseases. Pol II transcription is strictly regulated at three main stages: initiation, elongation, and termination by numerous regulatory factors, including kinases and phosphatases, chromatin structure, and antisense transcripts. Dysregulation of Pol II elongation and the production of antisense transcripts are associated with various diseases, including cancer, diabetes, cardiac and neurodegenerative disorders. Therefore, a better understanding of the fundamentals of the regulation of these processes is of paramount importance for improved diagnostic markers and therapeutic treatments. We investigate Pol II transcription regulation in the fission yeast Schizosaccharomyces pombe and human cells. Our research leverages integrated approaches, including biochemistry, cellular and molecular biology, classical genetics, and chemical genetics—a technique to sensitize a kinase to unnatural ATP analogs—combined with genomics and proteomics.
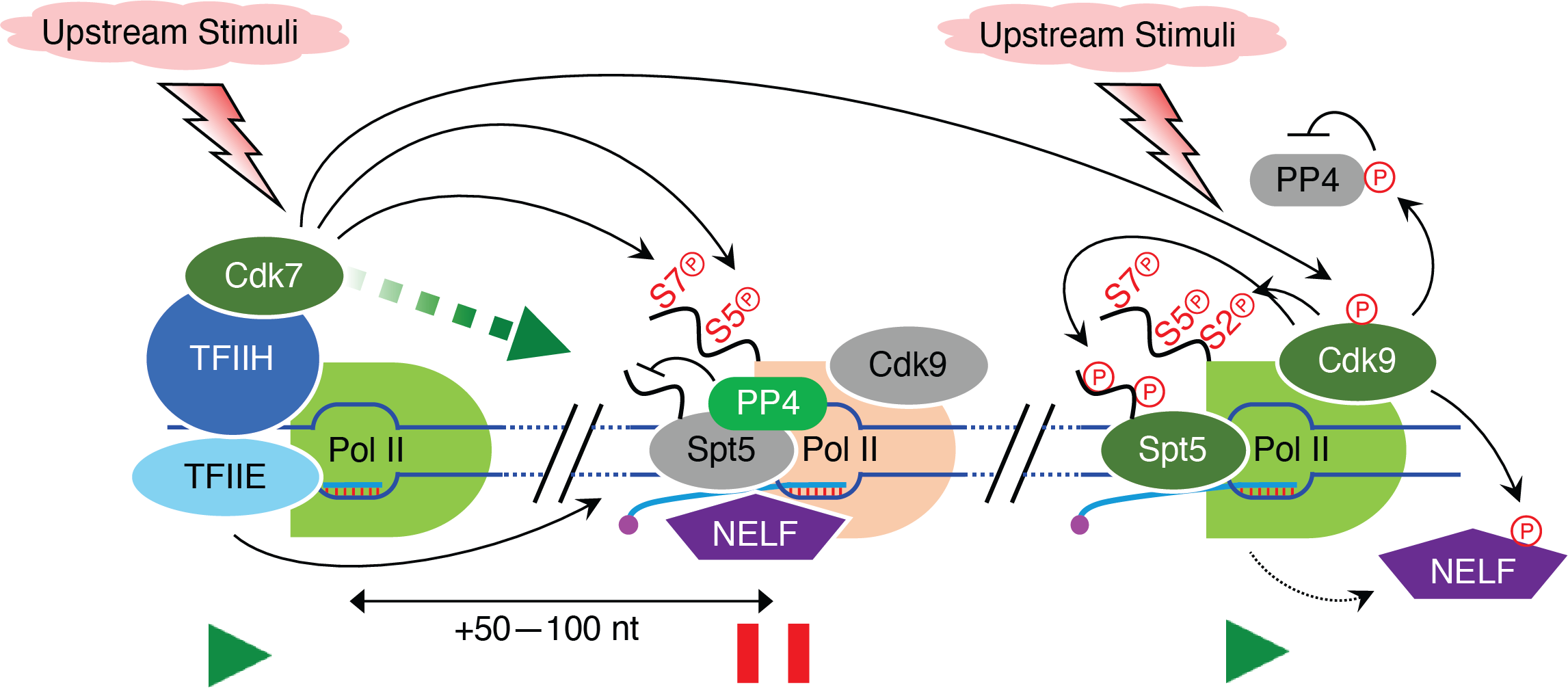
To obtain mechanistic insights into promoter-proximal pausing – Most of the genes in metazoan (and ~20% genes in fission yeast) are regulated by an early regulatory event, known as promoter-proximal pausing—Pol II is paused shortly after initiation around 20-80 nucleotides downstream of the transcription start site (TSS).Properly regulated release of stalled Pol II from the promoter-proximal pause site results in the synthesis of full-length transcripts. Mis-regulation of pausing or its release can result in abnormal gene expression. Given this early regulatory event’s decisive role in tuning Pol II transcription, dissecting the underlying molecular mechanisms is of utmost importance for understanding transcriptional homeostasis and its disruption in human diseases. Emerging studies suggest that the distinct kinase-phosphatase switch mechanisms control the phosphorylation of effector proteins, modulating the pause establishment, maintenance, and release. These critical kinase-phosphatase networks are mostly unknown and need to be identified and characterized precisely. We seek to investigate the regulation of promoter-proximal pausing in fission yeast and human cells to understand how the coordination between kinases and phosphatases ensures the pause establishment and synchronized release beneficial for healthy cells.
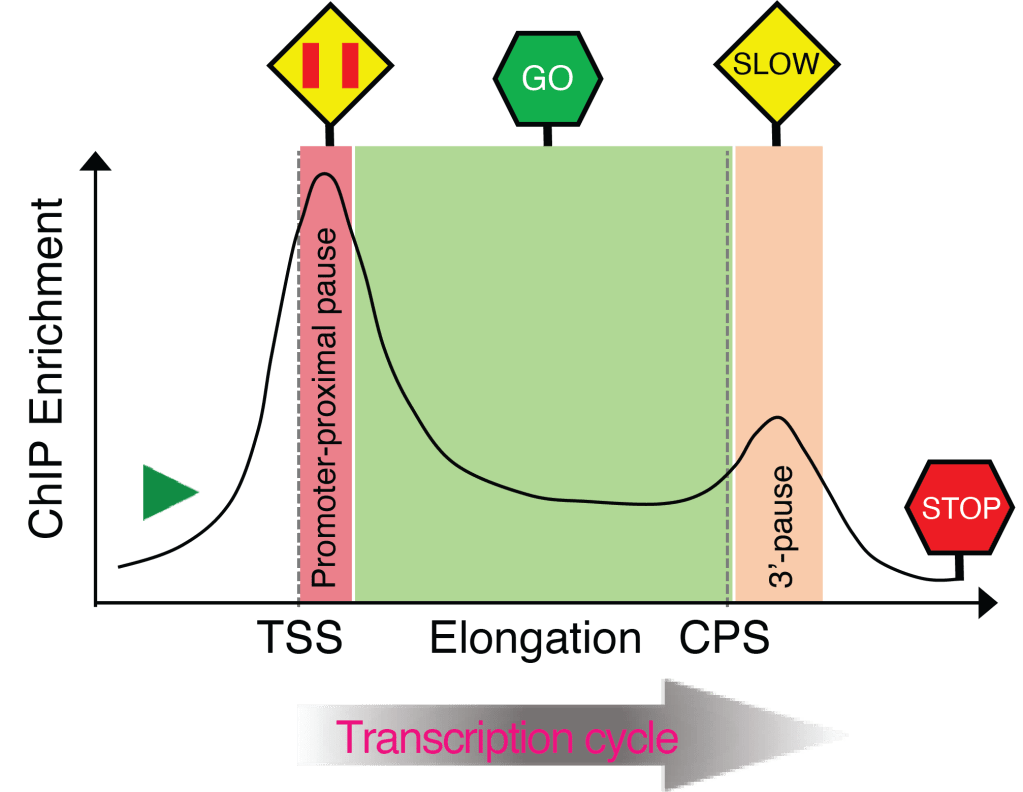
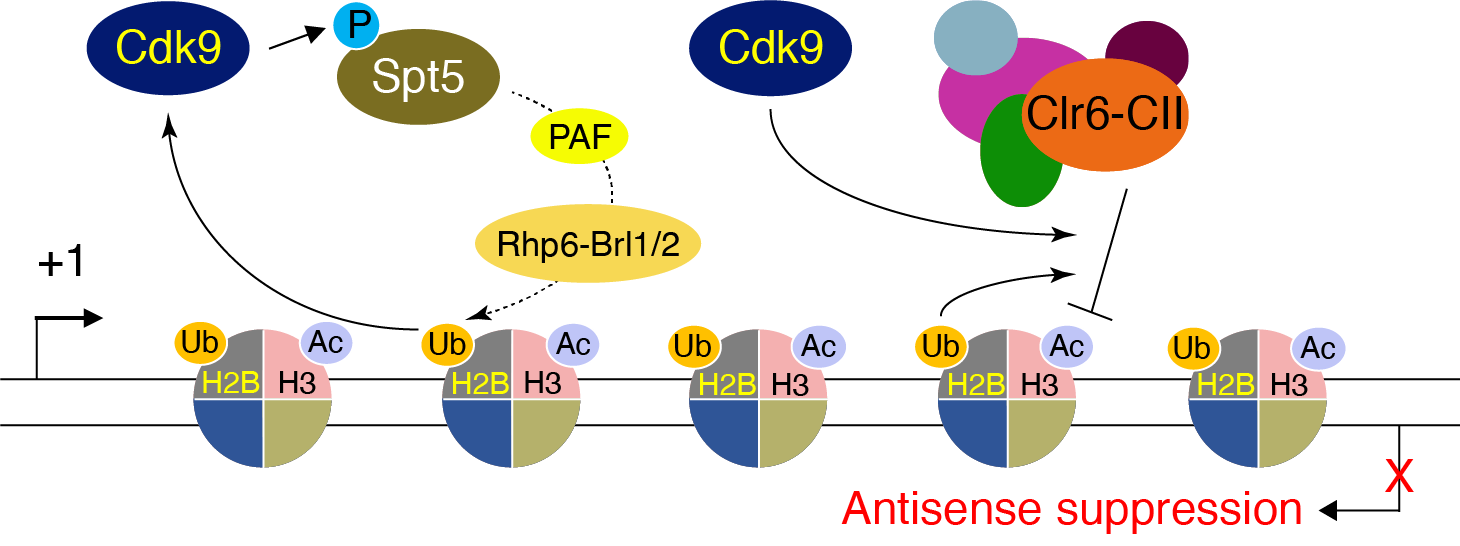
To investigate the coupling of transcription elongation and co-transcriptional processes – The variations in the rate of Pol II elongation have been implicated in controlling co-transcriptional processes such as 5′- and 3’-end processing, antisense transcription, alternative polyadenylation (APA), and splicing of pre-mRNA. However, much is still unknown, how the elongation rate is controlled and, consequently, the coupled process. The current hypothesis is that normal speeds of Pol II elongation favor the recruitment of factors necessary to execute a particular step, whereas slower Pol II promotes aberrant recruitment of factors, resulting in premature outcomes; conversely, faster rates impair the timely execution of exact steps. The primary objective here is to examine unidentified and uncharacterized connections among kinase-phosphatase antagonisms, rate of elongation, post-translational modifications (PTMs) of histones, pre-mRNA splicing, and transcription polarity.
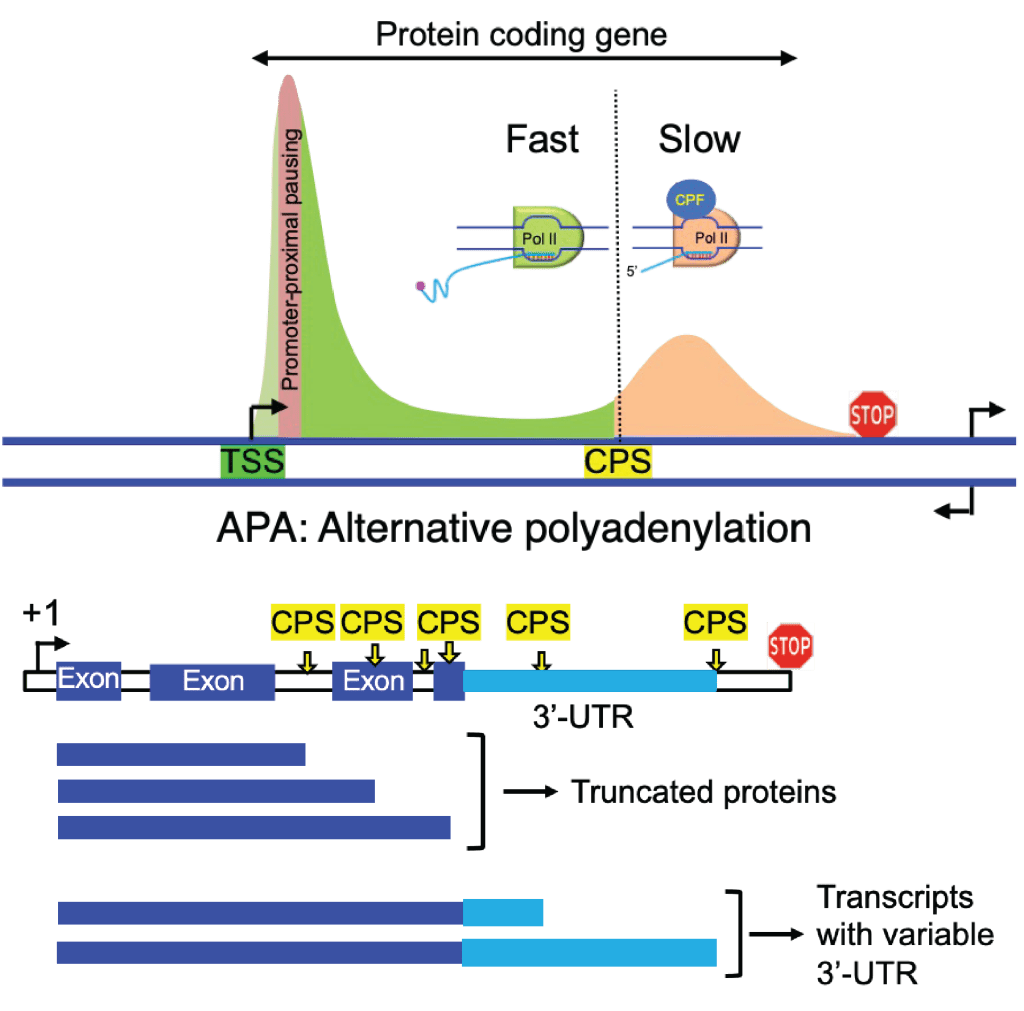
To uncover how spatial and temporal phosphorylation events influence termination – The elongation to termination transition, a crucial step near the end of transcription, prepares Pol II for efficient and accurate termination following a series of sequential events: (1) deacceleration of elongating Pol II while crosses the cleavage and polyadenylation signal (CPS), leading to (2) accumulation of Ser2 phosphorylation of Pol II carboxy-terminal domain (CTD), which in turn facilitates (3) the recruitment of factors involved in pre-mRNA 3’-end formation and termination. A long-standing puzzle was how the transition from elongation to termination is initiated. Recently we identified a novel bistable switch mechanism comprising cyclin-dependent kinase 9 (Cdk9) and protein phosphatase 1 (PP1) that rapidly reverses phosphorylation at the CTD of an essential elongation factor, Spt5 (and possibly other Cdk9 substrates) during the traversal of the elongation machinery through the CPS, leading to Pol II slowing. The Spt5 CTD phosphorylation is inversely correlated with Pol II CTD Ser2 and Thr4 phosphorylation at the 3’-end of genes. However, how their reciprocal relations functionally link to influence the termination remains less understood. We will assess the spatial and temporal connections of various phosphorylation events and characterize their molecular roles in transcription termination.



